BOTOX®
Approved by the FDA in 2002 for cosmetic use, BOTOX effectively treats glabellar lines (frown lines between the eyebrows), crow’s feet (lines at the outer corners of the eyes), and forehead lines (horizontal lines across the forehead). Additionally, trained aesthetic injectors often use BOTOX for off-label treatment areas. As the first neurotoxin approved for cosmetic use in the US, BOTOX remains the most popular choice for reducing the appearance of wrinkles.
The procedure is quick and relatively painless, with results lasting several months before gradually wearing off. At Pinnacle Dermatology, we specialize in using BOTOX to treat wrinkles, forehead lines, crow's feet, and frown lines between the eyebrows. Schedule your appointment today to achieve a more youthful look with BOTOX at Pinnacle Dermatology.
BOTOX Before and After Photos: Dr. Dino Mendez

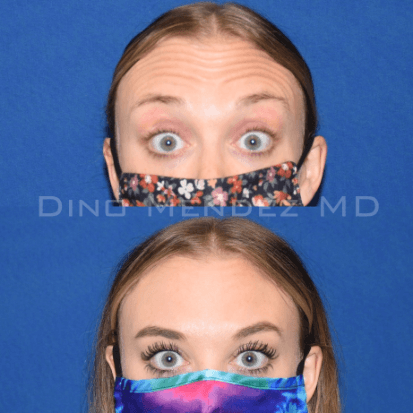

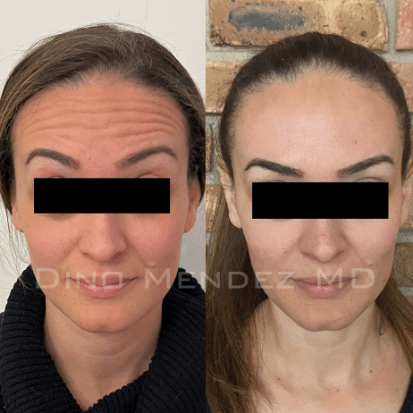
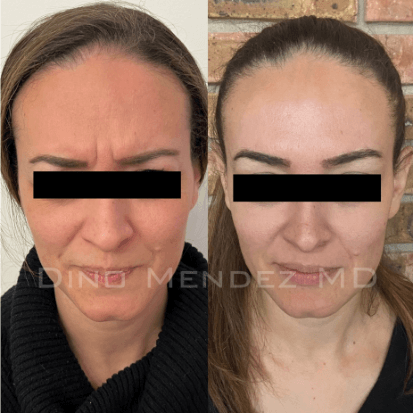



Benefits of BOTOX
- Wrinkle Reduction: Diminishes the appearance of fine lines and wrinkles, particularly on the forehead, around the eyes, and between eyebrows.
- Non-Invasive: A procedure that involves injections, offering quick results without significant downtime.
- Youthful Appearance: Provides a youthful and rejuvenated look by relaxing targeted facial muscles.
- Versatility: Addresses various concerns beyond wrinkles, such as excessive sweating (hyperhidrosis) or migraines.
Common Side Effects of BOTOX
- Temporary bruising at the injection site.
- Mild swelling.
- Redness.
- Temporary mild headache.
What to Expect at Your BOTOX Appointment
We begin by gently cleansing your face to clear away oil, makeup, and other debris. We then make a series of quick injections in or around your primary areas of concern. We may ask you to make certain facial expressions during this time to ensure accuracy. Each injection lasts only a moment and feels like a small, quick pinch. Afterward, we offer you a cold compress to help minimize swelling.
You can expect to be at the office for about 30 minutes. Immediately following your treatment, you can resume nearly all of your regular activities. However, it's important to avoid movements that require you to bend forward or lower your head for the remainder of the day. This allows the product to be distributed evenly for attractive, natural‐looking results.
BOTOX Maintenance Recommendations
After receiving BOTOX injections, it's essential to follow specific maintenance recommendations to optimize results and ensure safety. Here are some common guidelines:
- Avoid Rubbing or Massaging: For the first 24 hours after treatment, refrain from rubbing or massaging the treated area to prevent the spread of Botox to unintended muscles.
- Stay Upright: Maintain an upright position for about 4 hours post-injection. Avoid lying down or bending over excessively during this time to prevent the migration of BOTOX to unintended areas.
- Avoid Strenuous Exercise: Refrain from engaging in vigorous exercise or activities that increase blood flow and body temperature for 24 hours post-treatment, as this might disperse the BOTOX.
- Follow-Up Appointments: Attend any scheduled follow-up appointments recommended by your healthcare provider to assess results and determine if touch-up injections are needed.
- Be Patient: Results from BOTOX injections usually start to appear within a few days but may take up to two weeks for full effect. Be patient and allow the treatment time to take its full course.
BOTOX Maintenance Schedule
- The frequency of BOTOX treatments range between 3 and 6 months but may change based on various factors like metabolism and how active one's facial muscles are.
- Frequency also depends on your desired look resulting from the injections.
- It's best to consult with your dermatologist or skilled injector to determine the best treatment plan for you.
BOTOX FAQs
On average, BOTOX effects last around 3 to 4 months. After this period, the muscle activity gradually returns, and wrinkles may reappear. To maintain the desired results, individuals often undergo follow-up treatments. It's important to note that the longevity of BOTOX effects can be influenced by factors such as individual metabolism, the amount of product injected, and the specific area treated. Always consult with your provider for personalized information and recommendations.
When administered properly by a skilled and experienced skin care professional, BOTOX treatments are designed to provide natural-looking results. The goal is usually to soften wrinkles and fine lines while maintaining facial expressiveness. However, the outcome can depend on the specific technique used by the injector and the amount of BOTOX administered.
If too much BOTOX is injected or if it is not placed correctly, it can potentially lead to a "frozen" or "unnatural" appearance. This occurs when the muscles responsible for facial expressions are overly relaxed, limiting movement. To avoid this, it's crucial to choose a qualified and trained professional who understands facial anatomy and can tailor the treatment to your individual needs.
During the consultation, discuss your aesthetic goals with the provider and express any desires about wanting a natural look. A skilled injector can adjust the dosage and injection sites to achieve the desired outcome while preserving a natural range of facial expressions. It's important to follow the provider's recommendations and, if needed, schedule a follow-up appointment for any adjustments.
BOTOX and Dysport: Both are injectable neurotoxins used to reduce wrinkles. BOTOX (Allergan) and Dysport (Ipsen) have similar effects but differ in onset, spread, and cost. Results last about 3-4 months for both. Consult a dermatology professional for personalized advice.
BOTOX, or botulinum toxin, is considered safe when administered by a qualified and experienced dermatologist or certified injector. It has been used for various medical and cosmetic purposes for many years with a well-established safety record. However, like any medical procedure, there are potential risks and side effects.
Common side effects of Botox are typically mild and temporary, including bruising, swelling, and redness at the injection site. Rare but more serious side effects may include allergic reactions or unintended muscle weakness if the toxin spreads beyond the intended area.
The appropriate age to start BOTOX can vary based on individual factors such as genetics, skin condition, and personal preferences. BOTOX is commonly associated with addressing dynamic wrinkles, which are caused by repetitive muscle movements. While it's often used as a preventive measure for wrinkles, there is no strict age requirement.
Generally, individuals in their late 20s to early 30s may consider BOTOX as a preventive measure to minimize the development of fine lines and wrinkles. However, the decision to start BOTOX is highly individual and depends on factors such as:
- Visible Signs of Aging: If you have noticeable fine lines or wrinkles, regardless of age, BOTOX may be considered to address these concerns.
- Preventive Measures: Some individuals choose to start BOTOX earlier as a preventive measure to slow down the formation of wrinkles.
- Consultation with a Professional: It's crucial to consult with one of our certified dermatologists or injectors who can assess your skin and provide personalized recommendations based on your specific needs.
Ultimately, the decision to start BOTOX should be made in consultation with a healthcare professional who can evaluate your unique situation and help you determine the most appropriate timing for your cosmetic goals.
Learn More About BOTOX
How Pinnacle Dermatology Can Help
Featured Blogs
- Botox
- Cosmetic Treatments
Let's face it, we're all aging. BOTOX® can rejuvenate your face. Learn about the best locations for BOTOX® injections in this blog post.
Read More
- Botox
- Injectables or Fillers
- Cosmetic Treatments
Localized side effects have been associated with the SARS-CoV-2 mRNA vaccine in patients with a recent history of dermal filler injections.
Read More
- Botox
- Injectables or Fillers
- Cosmetic Treatments
Understanding the key differences between BOTOX® or fillers can help you make an informed decision on which treatment is right for you.
Read MoreFeatured Products

SkinCeuticals Phloretin CF Gel
Phloretin CF Gel is a revolutionary antioxidant technology combining 2% phloretin, 10% pure vitamin C (L-ascorbic acid), and 0.5% ferulic acid for enhanced protection against atmospheric aging – environmental damage and premature signs of aging caused by free radicals from UVA/UVB radiation. This daily topical vitamin C antioxidant also helps diminish the appearance of discoloration, refine skin texture, and improve skin tone. 1 fl oz / 30 mL

SkinCeuticals Triple Lipid Restore 2:4:2
Aging skin is increasingly susceptible to lipid depletion: the loss of natural compounds in skin's surface, present in the form of ceramides, natural cholesterol, and fatty acids. This lipid loss compromises skin's natural protective barrier. 1.6 fl oz / 48 mL


